SigzenPHARMA
Tailored ERP Solution for the Pharma Manufacturing Industry
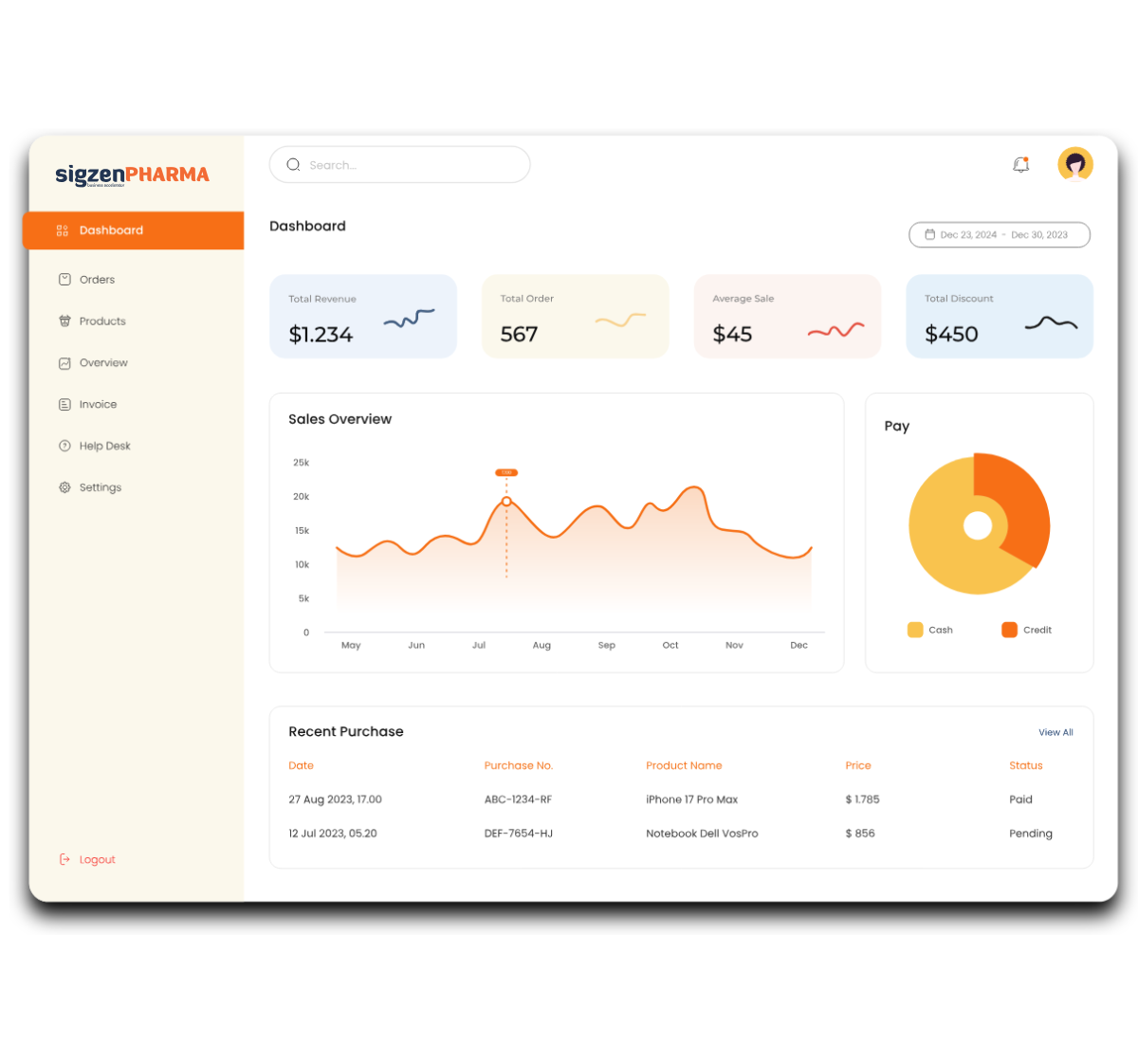
SigzenPHARMA is a purpose-built pharma ERP software solution developed by Sigzen Technologies for the pharmaceutical and Active Pharmaceutical Ingredient (API) manufacturing sector. Built on the powerful ERPNext and Frappe framework, this solution enables pharma businesses to seamlessly align with GMP, FDA, and 21 CFR Part 11 compliance requirements. Whether you’re an API manufacturer, bulk drug producer, or formulation-based plant, SigzenPHARMA provides the digital tools needed to optimize pharmaceutical operations, ensure batch traceability and drug traceability, and support regulatory audit readiness.
Designed with a deep understanding of regulated environments, SigzenPHARMA simplifies GMP-compliant documentation, improves data integrity, and automates the full production-to-quality lifecycle. With in-built support for batch manufacturing records, quality control protocols, validation, and real-time manufacturing analytics dashboards, this ERP is your partner for compliant growth in the pharmaceutical space.

Key Features
1. Regulatory Compliance (21 CFR Part 11 Ready)
SigzenPHARMA ensures regulatory adherence with advanced security and features.
Electronic Signatures
This feature enables secure sign-off on all records and transactions, ensuring accountability and compliance with digital signature requirements.
Audit Trail Logging
Every action performed within the system is meticulously tracked and logged, providing complete transparency and a verifiable history for all processes.
Access Control
The system uses role-based access to align with GxP standards, ensuring that personnel only have access to the data and functions necessary for their assigned roles.
Document Locking
Documents are made tamper-proof with version history, which prevents unauthorized changes and ensures the integrity of all critical records.
2. Digital Batch Manufacturing Records (BMR)
Digitally manage and monitor every stage of your manufacturing batch.
Batch-wise Tracking
Track every detail of your manufacturing process, from raw material input to final product output, with real-time status updates on each batch.
Stage-wise Log Sheets
Record essential data such as temperature, timings, operator information, and equipment usage at each stage of the manufacturing process for a comprehensive log.
Yield and Wastage Analysis
Easily compare planned outputs against actual results to analyze yield and wastage, helping you optimize production efficiency and reduce losses.
3. Pharma Quality Control & Quality Assurance
Ensure end-to-end QC integration across procurement, manufacturing, and dispatch.
Sampling and Inspection Templates
Use standardized templates for sampling and inspection, which can be applied to raw materials (RM), packaging materials (PM), finished goods (FG), and in-process checks.
Deviation & CAPA Management
The system allows you to flag non-conformities and automatically trigger corrective action and preventive action (CAPA) workflows to address quality issues promptly.
Release Workflows
Quality assurance (QA) hold and release processes are seamlessly integrated with production, ensuring that only compliant products proceed to the next stage.
4. Good Manufacturing Practice (GMP) Validation Docs
Ready-to-use templates and workflows to assist your compliance team.
Installation Qualification (IQ)
This module helps you validate the system and environment to confirm that they are installed correctly and ready for use.
Operational Qualification (OQ)
Conduct functional testing and SOP validation to verify that the system operates as intended, aligning with your standard operating procedures.
Performance Qualification (PQ)
Validate your processes with mock and live runs to ensure consistent performance and product quality under real-world conditions.
5. Inventory Control and Traceability in Pharma
Full visibility into materials at every stage of your supply chain.
Batch-wise Stock Management
Manage your materials by batch, from the moment they are received to their final dispatch, ensuring complete traceability throughout the entire supply chain.
Expiry and Retest Tracking
The system provides alerts and isolation workflows for materials nearing their expiry or retest dates, helping you manage stock effectively and prevent non-compliant materials from being used.
Cold Chain & Quarantine Zones
Define specific storage conditions and segregate materials based on their quality status, ensuring that sensitive products are handled appropriately and non-conforming items are quarantined.
6. Pharma Procurement Compliance & Vendor Management
Manage pharma procurement with compliance oversight.
Approved Vendor List (AVL)
Link vendors to specific materials.
Vendor Audit Logs
Attach qualification reports and keep a detailed log of all vendor audits, providing a clear record of vendor compliance and performance.
Purchase Inspection & Rejection
Integrate quality control into your procurement process by allowing for QC-based material acceptance and rejection, which ensures that only high-quality materials are used in production.
7. HRMS, Compliance Training & SOP Access Control
Ensure compliance at the personnel level.
Training Matrix
This module helps you track which employees have been trained for specific processes or equipment, ensuring that all operators are qualified for their tasks.
GMP Role Assignment
Control access to the system based on an employee’s competency and assigned GMP roles, which reduces the risk of unauthorized actions and data breaches.
Shift and Bio-Attendance
Efficiently manage multi-shift plant operations with a bio-attendance system that tracks employee hours and ensures accurate record-keeping.
8. Pharma ERP Finance & Batch Costing Module
Control costs and maintain financial clarity.
Batch Costing Ledger
Get a detailed breakdown of all costs associated with each production batch, including material, labor, and utility costs, for accurate cost analysis.
Multi-level Approvals
Implement multi-level approval workflows for all purchase and production spending, ensuring that financial controls are in place and expenditures are authorized.
Integrated GST & Compliance
The system is built to handle Indian tax laws and includes integrated GST and compliance features for seamless financial reporting and regulatory adherence.
Integration with Other ERPNext Modules
Enable seamless pharma operations through system-wide integration.
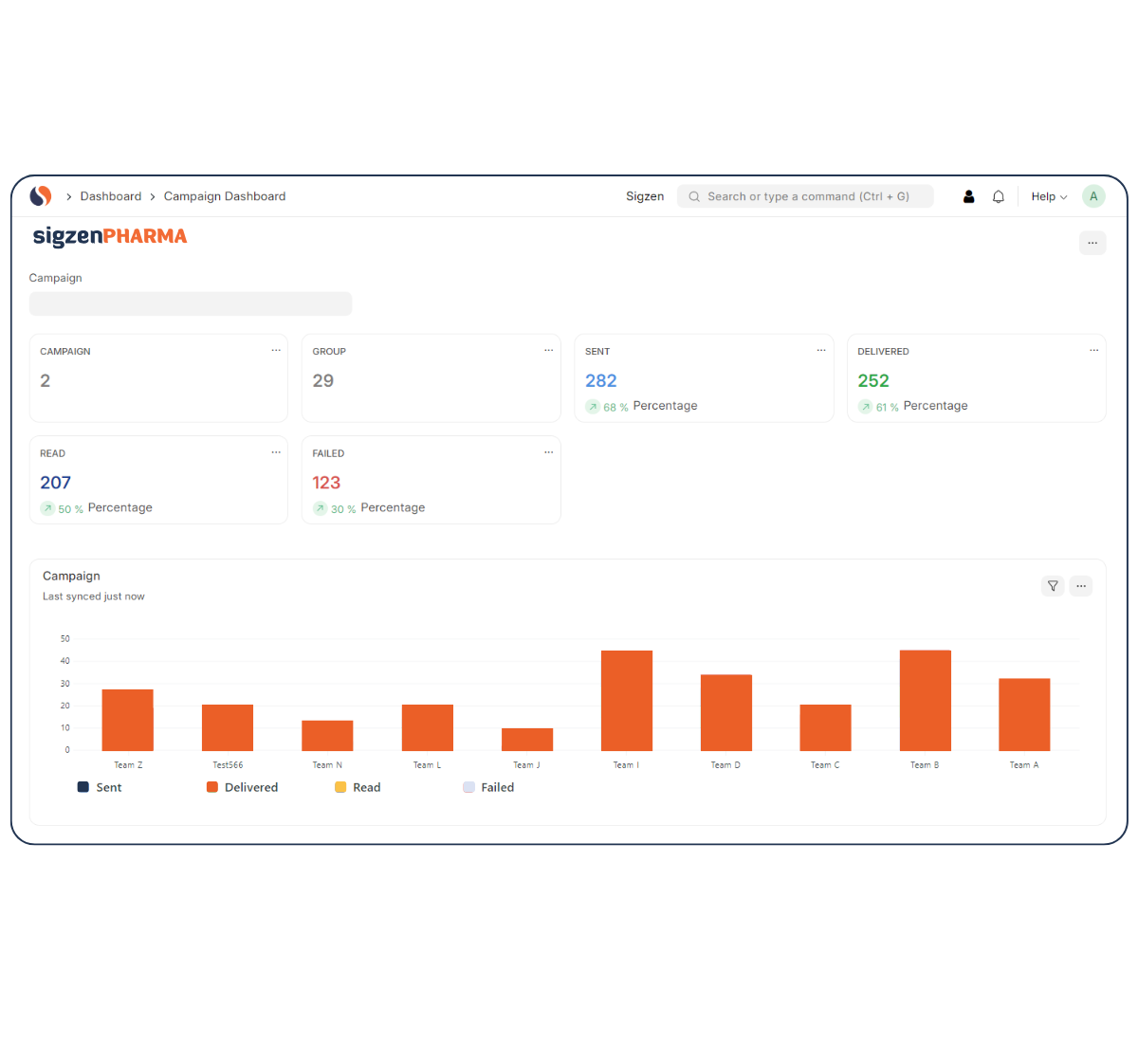
Case Studies
1. Digitizing Compliance for an API Manufacturer
Challenge
Paper-based BMR and quality logs delayed audits.
Solution
SigzenPHARMA replaced all paper trails with digital workflows.
Results
60% reduction in document errors, audit readiness in under 3 weeks.
2. Centralized Operations in a Multi-Plant Setup
Challenge
Fragmented procurement and inconsistent batch tracking.
Solution
Unified ERP with batch-wise stock movement and vendor compliance workflows.
Results
Increased batch yield accuracy and reduced procurement cycle time by 30%.

Tutorials & Guides
To help you get the most out of SigzenPHARMA, we provide the following resources:
Frequently Asked Questions
Is SigzenPHARMA compliant with 21 CFR Part 11 regulations?
Yes. SigzenPHARMA is fully compliant with 21 CFR Part 11. It includes electronic signatures, audit trails, and role-based access control, ensuring your pharmaceutical business meets GMP and FDA compliance requirements for digital documentation and quality systems. Our pharma software is specifically designed to adhere to these strict standards, providing you with a reliable and secure platform.
How does SigzenPHARMA handle multi-plant operations and batch scheduling?
SigzenPHARMA supports multi-company and multi-plant ERP configurations, allowing centralized control over batch manufacturing scheduling, inventory management, and procurement workflows across multiple sites. Our ERP software for pharmaceuticals provides a unified system for managing complex, multi-site operations with ease.
Can SigzenPHARMA integrate with external LIMS or lab weighing systems?
Yes. It offers seamless integration with LIMS (Laboratory Information Management Systems) and digital weighing systems using REST APIs and plug-ins, enabling automated test data capture and lab process synchronization. This capability makes our pharma ERP a central hub for all your lab and manufacturing data.
Does SigzenPHARMA simplify regulatory audits and inspections?
Absolutely. The system provides audit-ready reports, digital BMRs, and quality logs, all exportable for regulatory agencies like USFDA, MHRA, WHO-GMP, improving regulatory inspection readiness and compliance transparency. This streamlines the audit process, saving time and resources.
What type of user training and system validation support is provided?
SigzenPHARMA includes complete SOP-based training, IQ/OQ/PQ validation templates, and computer system validation (CSV) documentation, helping your QA teams validate systems and educate personnel efficiently. Our support ensures a smooth implementation and ongoing user proficiency.
Is batch costing and financial tracking available within SigzenPHARMA?
Yes. With the Pharma ERP Finance & Batch Costing Module, you can monitor material costs, labor charges, energy consumption, and cost variance for each production batch—fully integrated with GST compliance and multi-level approval workflows. This pharma software provides comprehensive financial visibility.
How does SigzenPHARMA ensure pharmaceutical inventory traceability?
It offers batch-wise stock management, expiry and retest tracking, and cold chain monitoring, supporting traceability from raw material to finished goods dispatch—crucial for pharma compliance and product recalls. This feature is vital for maintaining product safety and quality.
Can SigzenPHARMA manage GMP training and operator compliance?
Yes. Through the HRMS and Training Records module, it enables training matrix tracking, GMP-based role assignments, and shift bio-attendance logging, ensuring workforce compliance and audit preparedness. This helps you maintain a skilled and compliant team.
How does SigzenPHARMA enhance pharmaceutical quality control (QC)?
The system includes customizable QC sampling templates, in-process checks, and CAPA tracking, helping ensure consistent product quality and regulatory compliance across all manufacturing stages. This proactive approach to quality management reduces risks and improves product integrity.
Is cloud-based deployment and mobile accessibility supported?
Yes. SigzenPHARMA can be deployed on the cloud and accessed via mobile ERP dashboards, providing real-time visibility into production, inventory, and quality metrics, even while on the move. Our cloud ERP for pharma offers flexibility and remote access for your team.
Is SigzenPHARMA ready for 21 CFR Part 11 compliance?
Yes. It includes all required features like audit trails, electronic signatures, and access controls.
Is training and validation support included?
Yes. We provide SOP-aligned user training, IQ/OQ/PQ templates, and assistance.
Schedule a Free Pharma ERP Demo & Consultation
Discover how SigzenPHARMA can transform your pharmaceutical manufacturing business. Contact us today for a free demo and see how our can streamline your batch operations, ensure regulatory compliance, and improve plant efficiency. pharmaceutical ERP software solution.
Trust Elements
” From inventory to QC reports, everything is at our fingertips now. Extremely useful during customer audits.
Sanjay P.Formulation Plant Headbatch Traceability and Drug Traceability
” SigzenPHARMA helped us clear our regulatory inspection without a single note. Their BMR digitization and audit trails are brilliant.
Dr. Ramesh K.API Pharma